Monday, May 24, 2010
Mills Canyon Field Trip
Here's a map of the route to the park, Mr. Olson's Chemistry class went to. I did not go to the field trip, but looks like people had a lot of fun there and learned a lot of new stuff!
View Mills Canyon Field Trip Map in a larger map
Wednesday, May 19, 2010
Mini Project: making ice cream


photo credit: me
ingredients
- 1 cup of milk
- 1 tsp of vanilla
- 6 tbs of sugar
- rock salt
- ice
- small sandwich bag
- large sandwich bag
- pour the milk, vanilla, and sugar into the small sandwich bag
- zip it up
- fill half the bag of the big sandwich bag with ice and add a lot of salt rocks in there
- put the small sandwich bag with ingredients inside of it into the big one
- zip up the big one
- start shaking the bag
- shake for about 5-10 minutes or until ice cream starts to form
- you can add syrup or toppings to your ice cream if you want
- enjoy!
Mini Project: Defying Gravity


photo credit: me
materials
- a cup
- 1/2 cup of water
- piece of paper
- fill cup with water up to half way
- take piece of paper and put it over the cup
- slowly turn the cup upside down
- slowly remove your hand from the cup
- you will see the paper will not fall down
the paper was able to stay up because of the different air pressure inside and outside of the cup. The air pressure outside of the cup is higher and the pressure inside is lower. It made it able for the paper to stay in place, and defy gravity.
Gravity: the force of attraction by which terrestrial bodies tend to fall toward the center of the earth.
Mini Project : Play Dough



photo credit: Theresa Lee
IN CLASS WE MADE PLAY DOUGH! (:
ingredients we used
3 cups of all purpose flour
1 1/2 cup of salt
2 tbs of oil
food coloring ( if you want the play dough to have color)
directions
ingredients we used
3 cups of all purpose flour
1 1/2 cup of salt
2 tbs of oil
food coloring ( if you want the play dough to have color)
directions
- pour flour, salt, and oil into a big bowl
- mix
- add food coloring
- pour play dough into a dish that is able to be put on the burner
- have burner on high
- stir while on burner
- mixture should start to clump up and become play dough
- have fun!
Saturday, May 15, 2010
Chemistry Standard
Chem Standard 11g: Students know protons and neutrons have substructures and consist of particles called quarks.
A substructure is any basic structure or organization.
A quark is an elementary particle and a fundamental constituent of matter. Quarks combine to form composite particles called hadrons.
A hadron is a composite particle made of quarks held together by the strong force. Hadrons are categorized into two families: baryons ( made of three quarks), and mesons ( made of one quark and one antiquark). The best-known hadrons are protons and neutrons (both baryons), which can be found in the atomic nuclei. All hadrons except protons are unstable and undergo particle decay–however neutrons are stable when found inside the atomic nuclei. The best-known mesons are the pions and kaons, which were discovered during cosmic ray experiments during the late 1940s and early 1950s.
There are six types of quarks, known as flavors: up, down, charm, strange, top, and bottom. Up and down quarks have the lowest masses of all quarks. The heavier quarks rapidly change into up and down quarks through a process of particle decay: the transformation from a higher mass state to a lower mass state. Because of this, up and down quarks are generally stable and the most common in the universe, where as charm, strange, top, and bottom quarks can only be produced in high energy collisions.
The proton is a subatomic particle with an electric charge of +1 elementary charge. It is found in the nucleus of each atom, along with neutrons, but is also stable by itself and has a second identity as the hydrogen ion, H+. It is composed of three fundamental particles: two up quarks and one down quark.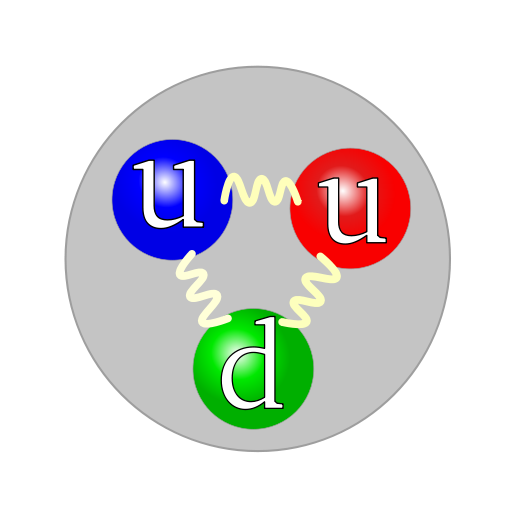
A quark is an elementary particle and a fundamental constituent of matter. Quarks combine to form composite particles called hadrons.
A hadron is a composite particle made of quarks held together by the strong force. Hadrons are categorized into two families: baryons ( made of three quarks), and mesons ( made of one quark and one antiquark). The best-known hadrons are protons and neutrons (both baryons), which can be found in the atomic nuclei. All hadrons except protons are unstable and undergo particle decay–however neutrons are stable when found inside the atomic nuclei. The best-known mesons are the pions and kaons, which were discovered during cosmic ray experiments during the late 1940s and early 1950s.
There are six types of quarks, known as flavors: up, down, charm, strange, top, and bottom. Up and down quarks have the lowest masses of all quarks. The heavier quarks rapidly change into up and down quarks through a process of particle decay: the transformation from a higher mass state to a lower mass state. Because of this, up and down quarks are generally stable and the most common in the universe, where as charm, strange, top, and bottom quarks can only be produced in high energy collisions.
The proton is a subatomic particle with an electric charge of +1 elementary charge. It is found in the nucleus of each atom, along with neutrons, but is also stable by itself and has a second identity as the hydrogen ion, H+. It is composed of three fundamental particles: two up quarks and one down quark.

Friday, April 16, 2010
PROPOSAL: MAKING CLOUDS
PRE LAB DISCUSSION
Removal of the stopper from an any opaque bottle or container results in the delayed evolution of a large cloud of water vapor some what like the mystical "jinni".
Removal of the stopper from an any opaque bottle or container results in the delayed evolution of a large cloud of water vapor some what like the mystical "jinni".
MATERIALS
SAFETY PRECAUTIONS
SCIENTIFIC PRINCIPLES
PROCEDURE

- Hydrogen Peroxide, H2O2
- Potassium Iodide, KI
- A large Bottle
- Stopper
- Rubber Gloves
- Tea Bag
- Safety Goggles
- Table Spoon
SAFETY PRECAUTIONS
- WEAR RUBBER GLOVES
- WEAR SAFETY GOGGLES
- CONTACT WITH HYDROGEN PEROXIDE MAY CAUSE BURNS
SCIENTIFIC PRINCIPLES
- Water Vapor is the gas phase of water
- Water Vapor is lighter or less dense than dry air
PROCEDURE
- Obtain a large opaque bottle and a stopper or cap to fit.
- Add 30-50 mL of 30% hydrogen peroxide to the bottle. CAREFUL: WEAR RUBBER GLOVES and SAFETY GOGGLES. WHEN HANDLING 30% HYDROGEN PEROXIDE. CONTACT WITH THE SKIN MAY CAUSE BURNS.
- Remove the tea from a tea bag and refill it with 1/4 table spoons of solid potassium iodide. Tie the tea bag securely with its string.
- Place the tea bag inside the neck of the bottle. While holding the string/ insert the stopper to hold the bag in place.
- When ready to perform the demonstration/ remove the stopper and allow the tea bag to fall into the hydrogen peroxide. Be sure to point the neck of the bottle up and in a safe direction.
- Within a few seconds, an exothermic reaction in the bottle will produce a large amount of oxygen gas and water vapor that will form an impressive cloud as it escapes from the bottle.

Friday, March 26, 2010
GOT THE 20 PT QUESTION ALL RIGHT!

WOOT WOOT!!
GOT THE 20 PT QUESTION ON THE CHEM TEST ALL RIGHT (; IM HELLLLLLUH SMART. HAHA jk
-____-
i got the 20 pt question all right, but failed on the multiple choice T.T
GOT THE 20 PT QUESTION ON THE CHEM TEST ALL RIGHT (; IM HELLLLLLUH SMART. HAHA jk
-____-
i got the 20 pt question all right, but failed on the multiple choice T.T
Thursday, March 11, 2010
SPRING BREAK PLANS!

SO I'M HAPPY TO SAY THAT I KNOW WHERE IM GOING FOR SPRING BREAK!! (:
LAS VEGAS!!!
i will be going with my cousins!! its going to be so fun!
the down side is that im paying for everything BY MYSELF. gah im going to be broke! the plane ticket was $230 two ways...and my mom paid $130 for it, i paid the other $100...then i have to pay for the hotel room which is $560 but we're splitting it between 4 people so i only have to pay $140 for 5 days..what a deal huh?? (: so, so far thats $240 for me...then i have to pay for my own food & buy my own stuff if i want to...so im still debating how much money to bring!!
i will be going to VEGAS on April 1-5 (: AND I GET TO SKIP ONE DAY OF SCHOOL! (: YAY
LAS VEGAS!!!
i will be going with my cousins!! its going to be so fun!
the down side is that im paying for everything BY MYSELF. gah im going to be broke! the plane ticket was $230 two ways...and my mom paid $130 for it, i paid the other $100...then i have to pay for the hotel room which is $560 but we're splitting it between 4 people so i only have to pay $140 for 5 days..what a deal huh?? (: so, so far thats $240 for me...then i have to pay for my own food & buy my own stuff if i want to...so im still debating how much money to bring!!
i will be going to VEGAS on April 1-5 (: AND I GET TO SKIP ONE DAY OF SCHOOL! (: YAY
IM SO HAPPPPPY! >:)

so BASICALLY.. there is no school on FRIDAY! yes!
& today which is a thursday is going to seem like a friday, and friday is going to seem like a saturday! how awesome is that?!!! & then on SATURDAY! IM GOING TO SIX FLAGS DISCOVERY KINGDOM WITH MY COUSINS!! very excited for it!! (:
CHINATOWN -____-

LOL, I'VE BEEN VISITING CHINATOWN A LOT LATELY. aha IM SO ASIAN! (: anyways me and cousin literally walked the whole chinatown, since we dont have anything else to do. there are actually really cool things in chinatown & everything is so cheap too! so i love it!
omg chem packets are killing me!!
SOOO LATELY...IN CHEMISTRY WE'VE BEEN HAVING A LOT OF PACKETS TO DO!! IT'S FREAKEN CRAZY!! AHA so little time to do so much >:O NOT COOL! ughh i dont like those packets...ohwell what can i do about it..at least im getting good grades on them (: so its okay I GUESS. my last packet that i got was about...
THERMOCHEMISTRY
the chemistry of energy!!
energy (Q=MCdeltaT)
the chemistry of energy!!
energy (Q=MCdeltaT)
SF A LOT LATELY

so...i've been going to sf a lot lately. just hanging out with my cousin & friends. so yeahhh. we walked all over the place, and did a lot of shopping. i didnt really get anything, since i didnt find anything that i really liked. i <3 SAN FRANCISCO! (:
Thursday, February 18, 2010
Friday, February 12, 2010
Sunday, February 7, 2010
SCHOOOOOOL.

schooool's been alright I GUESS...i mean..homework is a PAIN IN THE BUTT!
and, um trying to keep my grades up is pretty hard to do to!
i wish we didn't have homework. ugh what's the point? LOL.
we should have more days off at school. AND IM GLAD WE THE SOPHOMORES ARE DONE WITH THE CAHSEE.
i hope i passed.
Thursday, January 7, 2010
Like Dissolves Like
HELLLLLLLLLLO..ugh.
To DISSOLVE is when a chemical spreads evenly into another chemical.

A SOLUTE gets dissolved.
A SOLVENT does the dissolving.
"like dissolves like" is an expression used by chemists to remember how some solvents work. It refers to "polar" and "nonpolar" solvents and solutes. This means that substances are more likely to dissolve in other substances with similar chemical properties. polar solutes dissolve in polar solvents. Nonpolar solutes dissolve in nonpolar solvents.




example: Water (H2O) is polar. Oil is non polar. Water (H2O) will not dissolve oil. Water (H2O) is polar. Salt (NaCl) is polar. Like dissolves like so polar dissolves polar so water dissolves salt.
To DISSOLVE is when a chemical spreads evenly into another chemical.

A SOLUTE gets dissolved.
A SOLVENT does the dissolving.
"like dissolves like" is an expression used by chemists to remember how some solvents work. It refers to "polar" and "nonpolar" solvents and solutes. This means that substances are more likely to dissolve in other substances with similar chemical properties. polar solutes dissolve in polar solvents. Nonpolar solutes dissolve in nonpolar solvents.




example: Water (H2O) is polar. Oil is non polar. Water (H2O) will not dissolve oil. Water (H2O) is polar. Salt (NaCl) is polar. Like dissolves like so polar dissolves polar so water dissolves salt.
Subscribe to:
Comments (Atom)




